2026-03-09
Great Bay Bio’s CHO-K1 catalog medium AlfaMedX-K1 has successfully completed Drug Master File (DMF) filing with the U.S. Food and Drug Administration (FDA). DMF Number: 42995. This milestone marks another significant achievement in Great Bay Bio’s global expansion and commercialization journey, following the company’s ISO 9001:2015 certification.
With the DMF filing, AlfaMedX-K1 provides practical advantages for global customers: when used in FDA regulatory submissions for new drug applications, the DMF number can be directly referenced, greatly shortening product review and evaluation time, simplifying submission packages, and accelerating drug approval and market entry.

▲ AlfaMedX-K1, a CHO-K1 Catalog Medium, Completes FDA DMF Filing
AlfaMedX-K1, developed on Great Bay Bio’s AlfaMedX platform, leverages AI-driven analysis and predictive modeling of extensive experimental datasets to optimize medium composition and culture conditions. This catalog medium is specifically designed for CHO-K1 cell lines, supporting enhanced cell growth and improved protein expression.
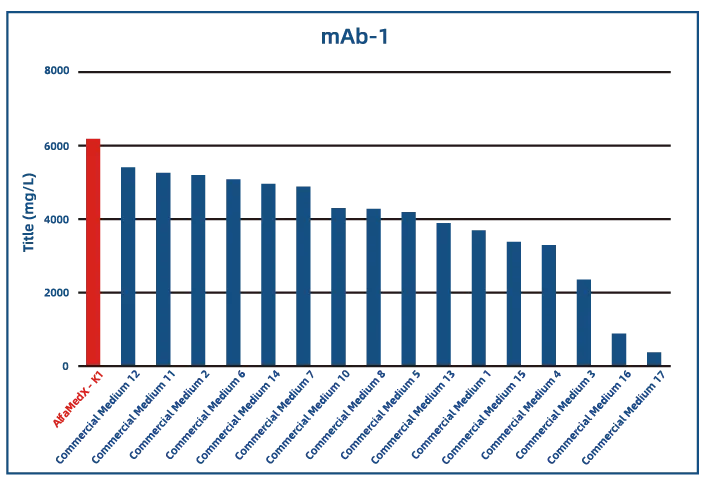
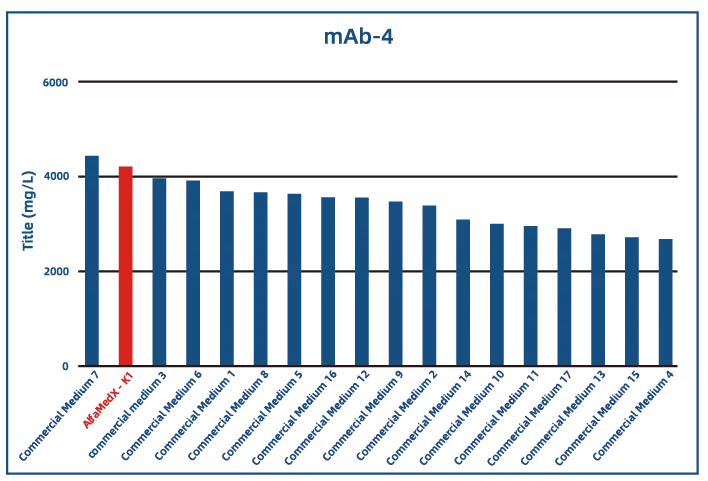
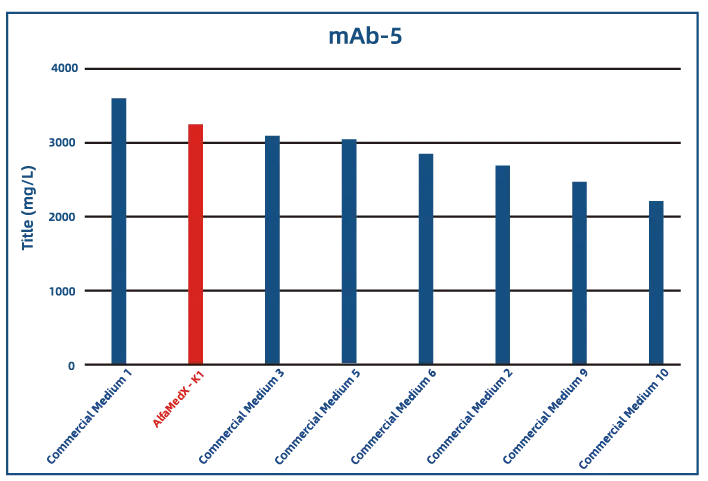
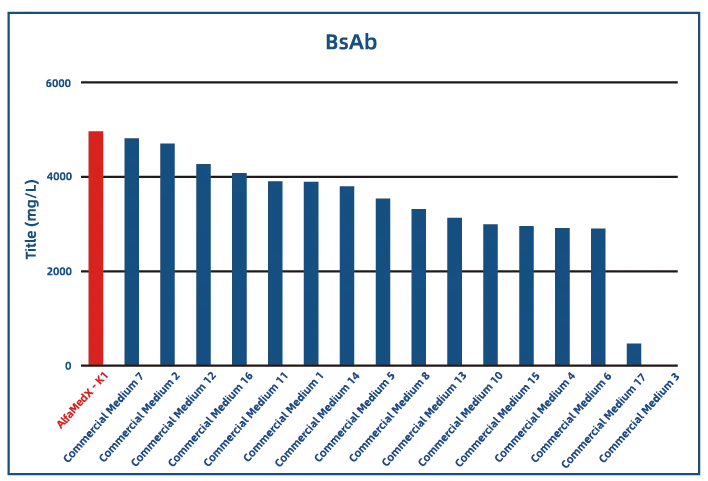
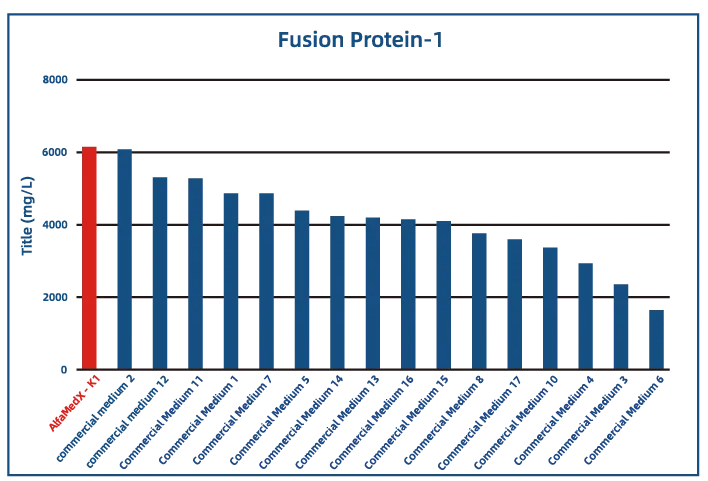
Comparative experimental data show that AlfaMedX-K1 demonstrates core advantages in CHO-K1 cell culture. When benchmarked against 17 commonly used commercial media, it consistently delivers excellent expression performance and broad adaptability across cell lines producing monoclonal antibodies, bispecific antibodies, and fusion proteins.
· Monoclonal Antibody (mAb)
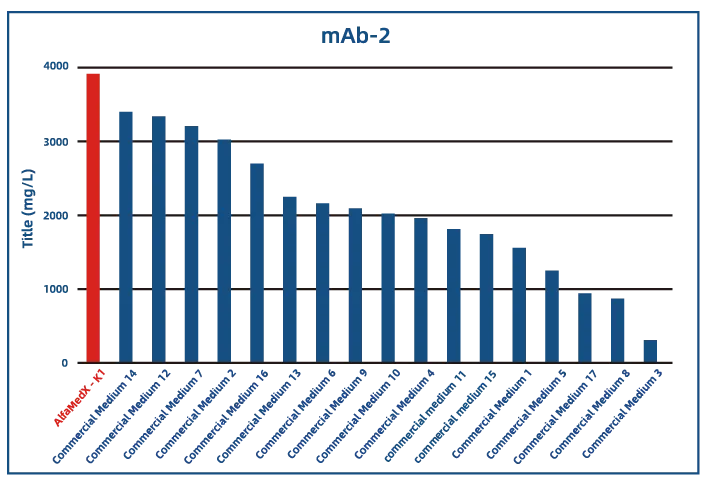
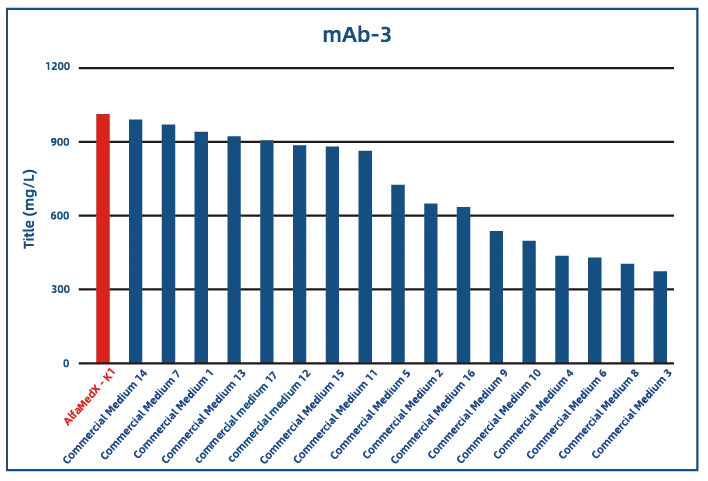
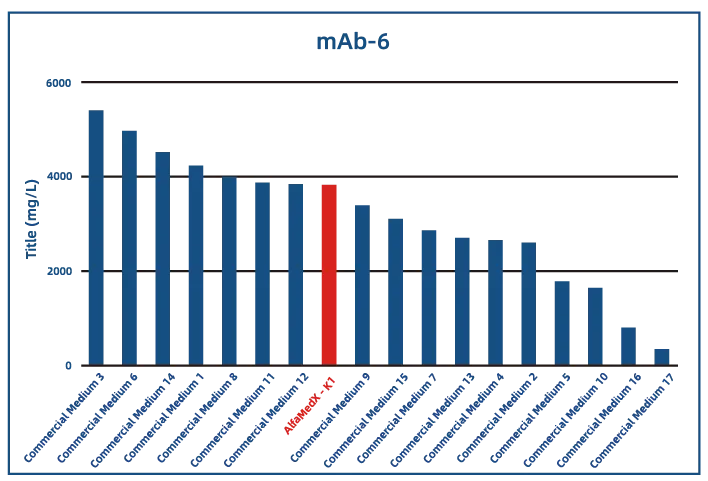
·Bispecific Antibody (BsAb)
· Fusion Protein
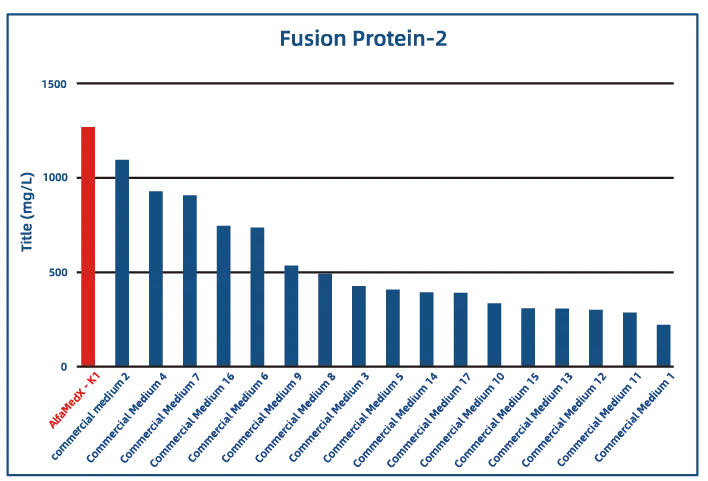
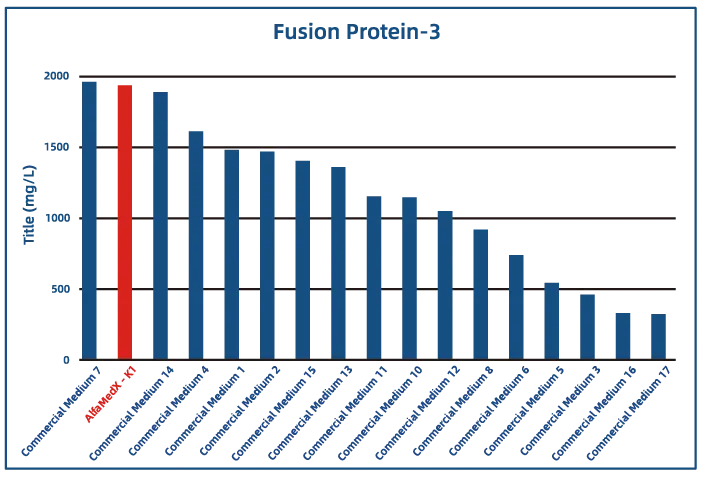
Key features for CHO-K1 cell lines (growth factor–free):

Great Bay Bio’s AlfaCell Delivers Average Titer Above 6.9 g/L Across Client Projects, with RCB Completed in Just 1.5 Months
2026-03-27

Great Bay Bio Recognized in the Deloitte Greater Bay Area Technology Fast 40 & Rising Star Program, Receiving Further Authoritative Recognition for AI-Enabled Bioprocess Innovation
2026-03-26

Kingsley Leung, Chairman of Great Bay Bio, Awarded the FHKI X OCBC Hong Kong Innopreneur Awards 2025, Recognizing Outstanding Innovation
2025-11-14