2026-03-27
Great Bay Bio’s site-specific integration cell line development platform, AlfaCell, has recently completed a new batch of client projects and delivered another strong set of results:
These results further demonstrate AlfaCell’s ability to support multiple molecule formats on a single platform, delivering stable, high titer without being limited by molecular complexity.
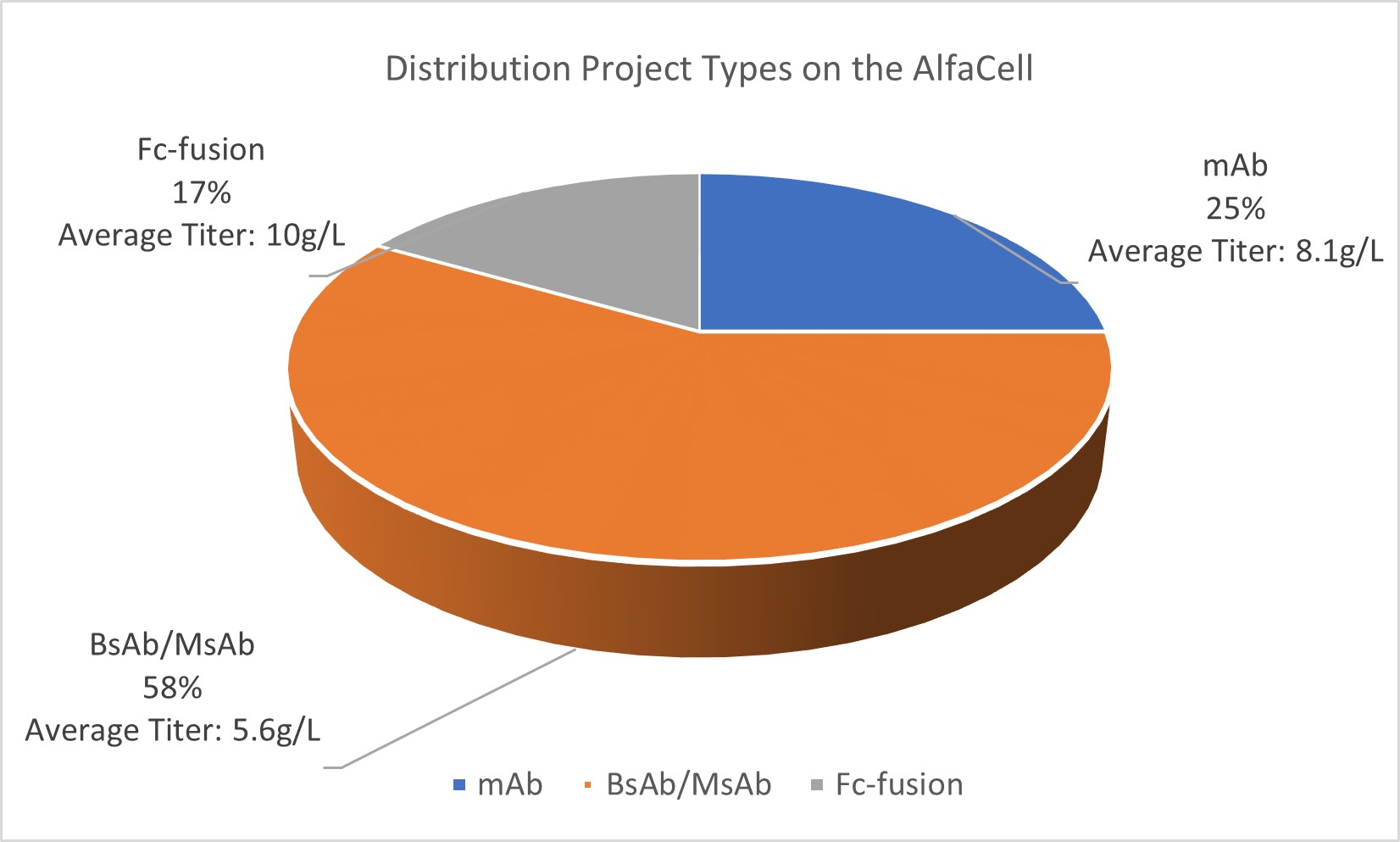
▲Distribution Project Types on the AlfaCell
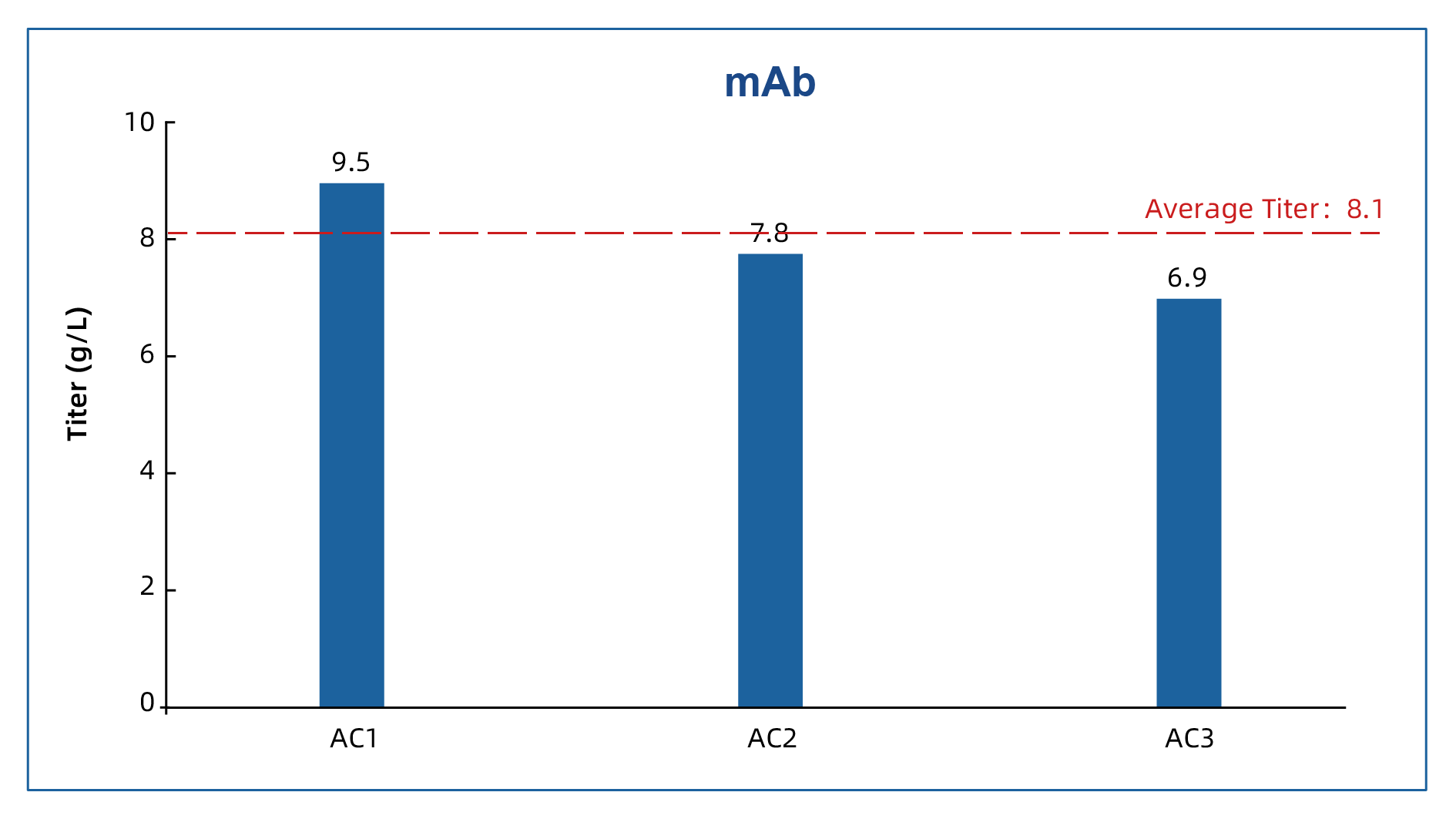
1. Monoclonal antibodies: average titer above 8 g/L
In three recent monoclonal antibody projects — AC1, AC2, and AC3 — fed-batch titers reached 9.5 g/L, 7.8 g/L, and 6.9 g/L, respectively, with an average titer of 8.1 g/L.
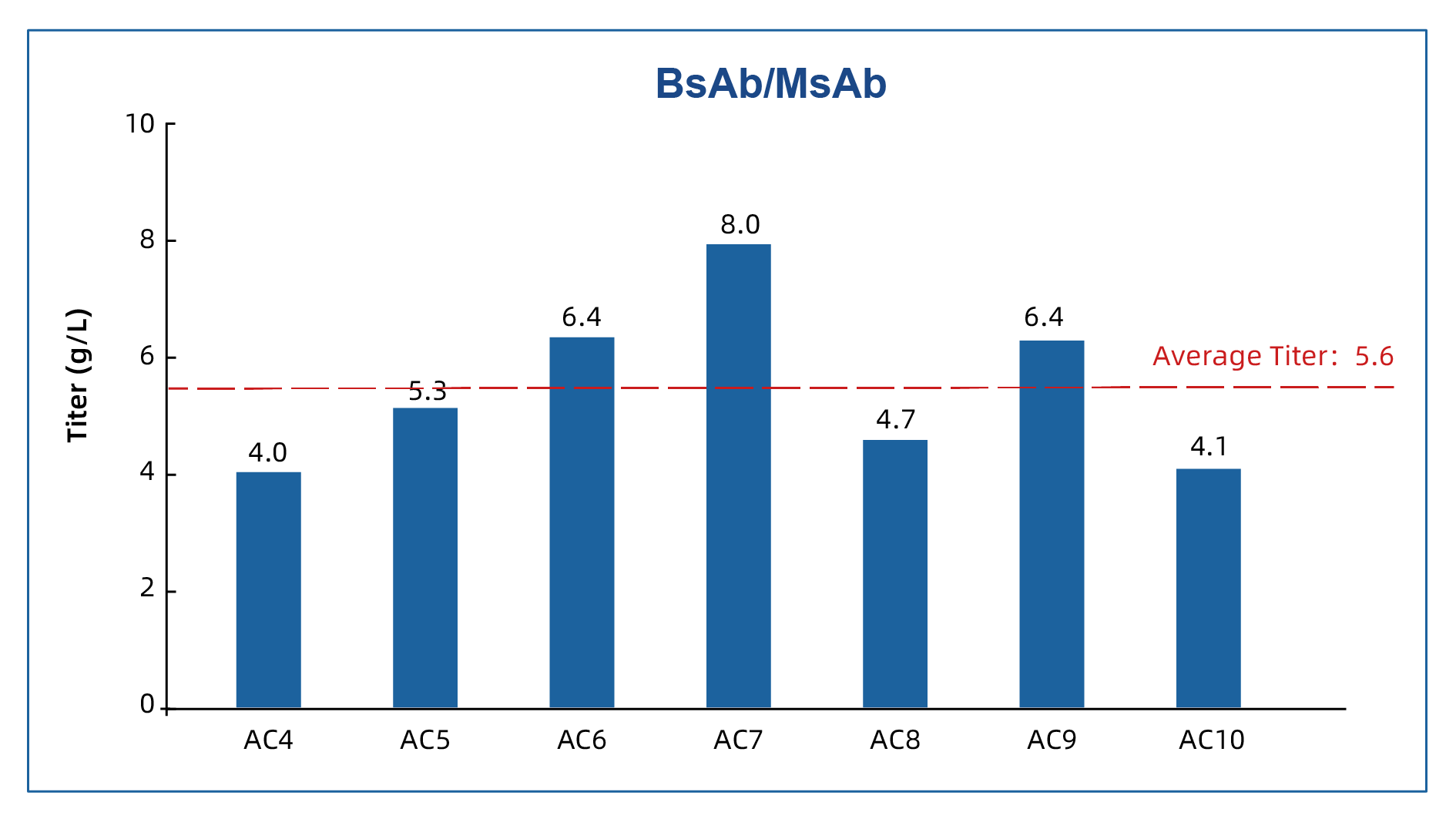
2. Bispecific/multispecific antibodies: average titer of 5.6 g/L
Across 7 projects involving symmetric bispecifics, three-chain bispecifics, and two-chain multispecifics, all achieved fed-batch titers above 4 g/L. Notably, the three-chain bispecific project A6 reached 6.4 g/L.
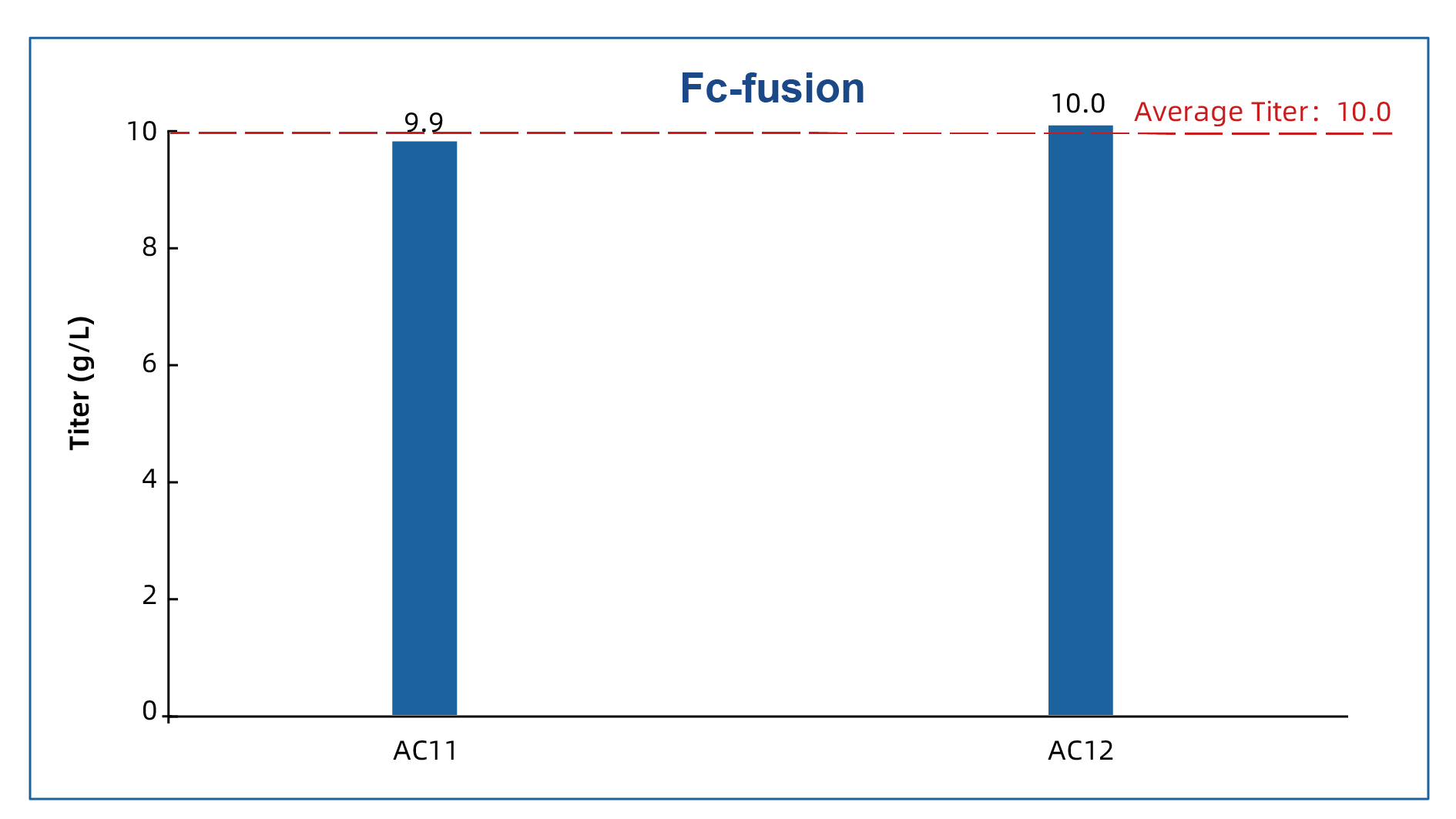
3. Fc-fusion proteins: titers reaching 10 g/L
Two recent Fc-fusion protein projects achieved fed-batch titers of 9.9 g/L and 10.0 g/L, respectively, with an average titer of 10.0 g/L.
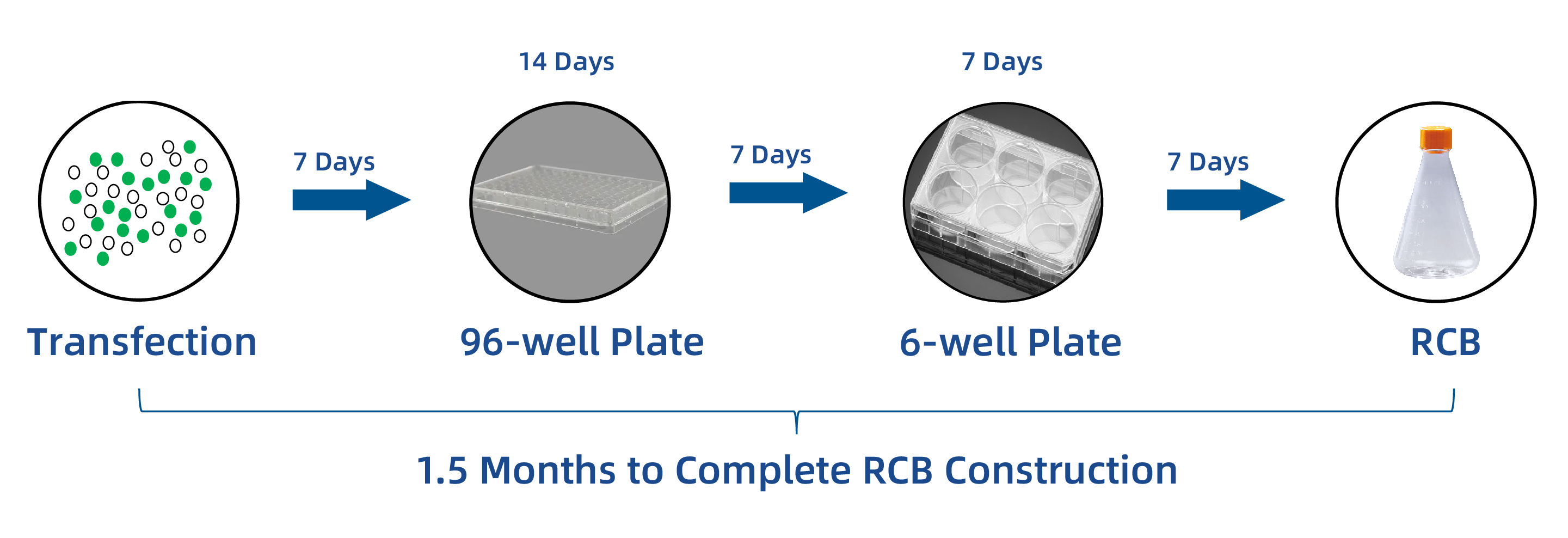
As the world’s first commercialized site-specific integration cell line development platform, AlfaCell supports the development of diverse product modalities with stable, high-titer cell lines and can complete the work that traditionally takes around 6 months to obtain an RCB in just 1.5 months.
AlfaCell demonstrated a strong positive correlation between 6-well batch culture performance and fed-batch expression levels, confirming that the platform not only delivers high titer but also ensures stable scalability, laying a solid foundation for both biologics development and industrial-scale manufacturing.
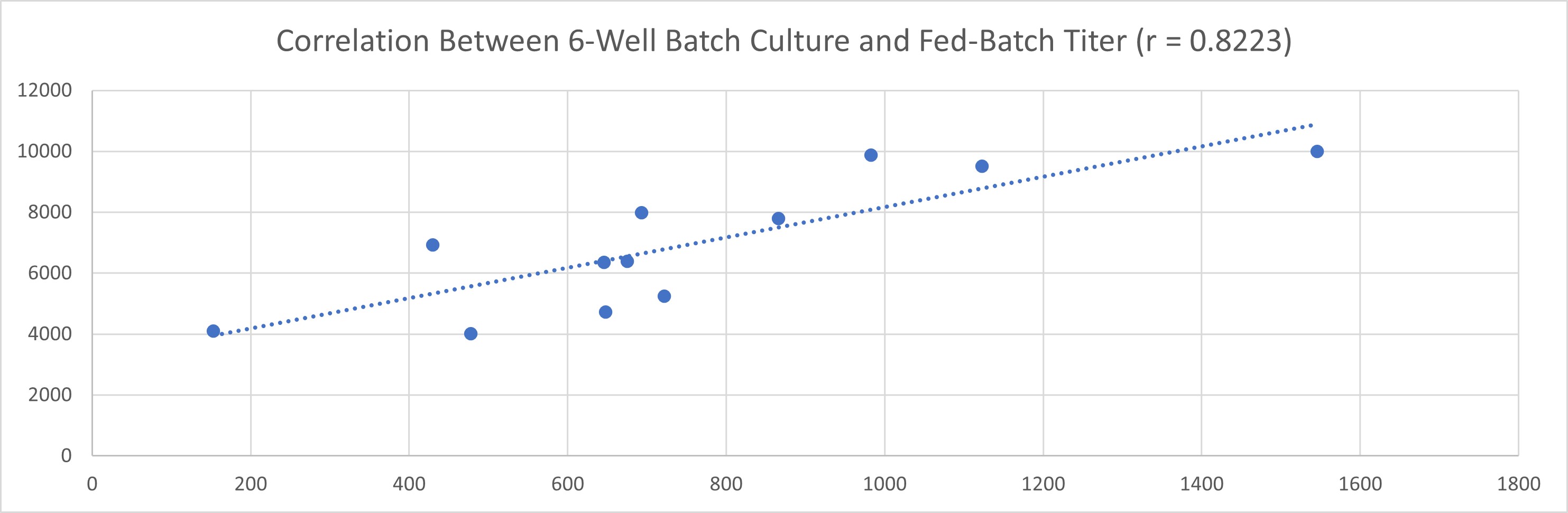
▲ Pearson correlation coefficient: r = 0.8223, indicating a strong positive correlation
Across 12 recent AlfaCell client projects, AlfaCell supported a broad range of product modalities, from well-established monoclonal antibodies to structurally complex bispecifics, multispecifics, and fusion proteins, all achieving the development goals of high titer and stable production. This strong set of data further validates AlfaCell’s technical strength as a platform that can support multiple product types, deliver stable high expression, and perform consistently across diverse molecular formats. Looking ahead, Great Bay Bio will continue to advance innovation in cell line development with AlfaCell at its core, providing global biopharma companies with more efficient, more stable, and better-adapted upstream development solutions, accelerating the R&D and industrialization of biologics, and making global bioprocessing simpler and more efficient.

Great Bay Bio Recognized in the Deloitte Greater Bay Area Technology Fast 40 & Rising Star Program, Receiving Further Authoritative Recognition for AI-Enabled Bioprocess Innovation
2026-03-26

Great Bay Bio’s CHO-K1 Catalog Medium AlfaMedX- K1 (Growth Factor–Free) Achieves FDA DMF Filing
2026-03-09

Kingsley Leung, Chairman of Great Bay Bio, Awarded the FHKI X OCBC Hong Kong Innopreneur Awards 2025, Recognizing Outstanding Innovation
2025-11-14